|
7/7/2023 0 Comments Periodic table quantum numbers 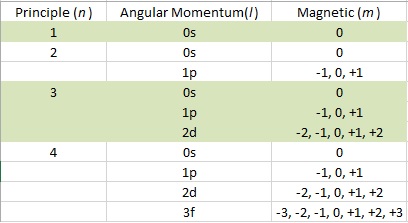 
So just use the orbital designation for quantum number shorthand. Just using the n and ℓ we call this "orbital" a 1s orbital. which is the 13th atom in the periodic table, so atomic number Z 13 and. The quantum numbers for this are n=1, ℓ=0, m ℓ=0, and m s=+1/2. Given an orbital, the quantum numbers that describe that orbital can be. So let's imagine a hydrogen atom in its lowest energy state. Identify the block in the periodic table to which the group 2 elements belong. The angular momentum quantum number can be used to give the shapes of the electronic orbitals. The s correlates to 0, p to 1, d to 2, and f to 3. The orbital letters are associated with the angular momentum quantum number, which is assigned an integer value from 0 to 3. In this notation we simply state the principal quantum number n as a number.Ī letter is used to denote the ℓ term as letters s,p,d, and f. This is a graphic representation of the 4fz3 electron orbital. And really, just the first two are needed to identify the right KIND of orbital and energy level. When they are paired in an orbital you show both together as a pair, ⥮.Īs stated in the list above, we tend to just say "orbital" when referring to a specific set of quantum numbers. Refers to orbital Possible values are ml 0, 1, 2, 3 No reference to periodic table but orbitals are drawn as diagrams (a box represents each orbital). In electron energy diagrams we use and up arrow or better, a "harpoon" ↿ to depict a +½ spin state and a down harpoon ⇂ to depict a –½ spin state. For lack of a better term, "spin" was the winner for the name of this property. Janets periodic table can be organized into rows representing electronic quantum numbers and. No two electrons can have an identical set of quantum numbers according to the Pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given state and therefore give insight into the building up of the periodic table of the elements.It was discovered much later because electrons were found to have another property that wasn't accounted for originally. Quantum Numbers, Hydrogen Atom In the solution to the Schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin. The Pauli exclusion principle (Wolfgang Pauli, Nobel Prize 1945) states thatno two electrons in the same atom can have identical values for all four of their quantum numbers. An electron can spin in only one of two directions (sometimes called up and down). The equation for each of the three variables gives rise to a quantum number and the quantized energy states of the atom can be specified in terms of these quantum numbers. Specifies the orientation of the spin axis of an electron. :max_bytes(150000):strip_icc()/AlkaliMetals-56a12cd73df78cf772682671.png)
For example, the electron configuration of helium (He), is 1 s 2 - the principal quantum number is. Separated into equations in terms of the spherical coordinates The principal quantum number tells us which principal electron shells the electrons occupy. The hydrogen atom solution to the Schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. The elements of the periodic table are sorted by their elemental properties, defined by the number of protons in the nucleus and the bonds formed by their electron structures. Quantum numbers for hydrogen atom Geometry of Hydrogen Atom Solution 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
